Drone Delivery
Researchers Evaluate the Delivery of Medicines Using Drones
Researchers Evaluate the Delivery of Medicines Using Drones
Drones have proved to be a promising and life saving tool as emergency responders in medical emergencies like cardiac arrest for example. Recently scientists Michelle Sing Yee Hii, Patrick Courtney and Paul G. Royall, with support from the Institute of Pharmaceutical Science, Department of Pharmacy, Faculty of Life Sciences & Medicine, King’s College London completed a set of tests to study the impact of drone transportation on the quality of a medicine.
In recent years, there is an increasing interest in drone delivery of medicines to remote areas. However, little has been reported about the feasibility and impacts of drone delivery on medicines, hence the need for this study.
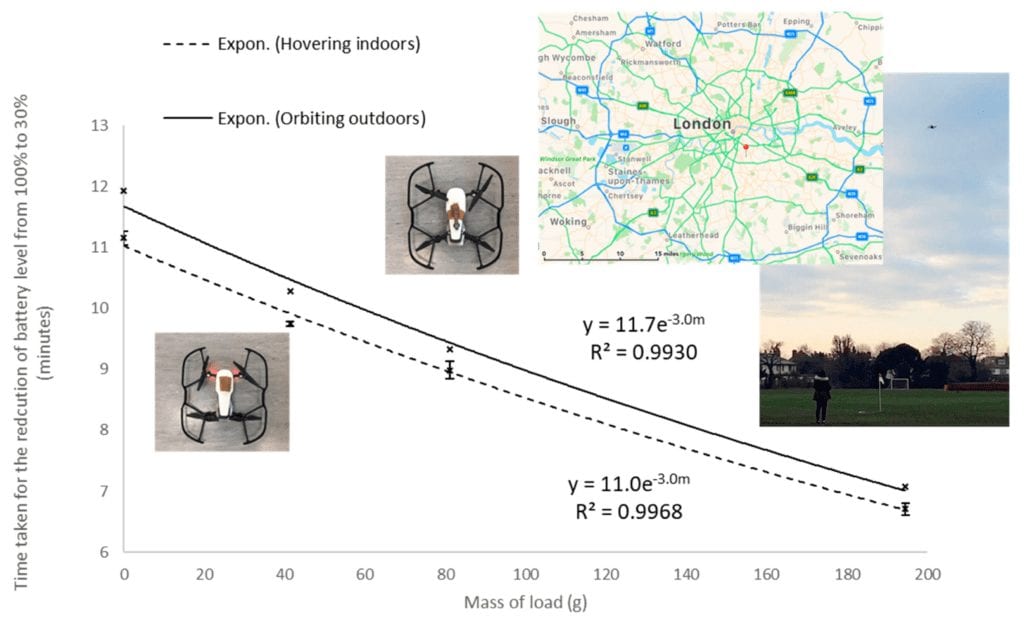
Time taken for the reduction of battery level from 100% to 30% (min) plotted against m (mass of load in kg). The exponential regression for drone hovering indoors: time = 11.0 e−3.0m (R² = 0.9968). Data points shown are the mean of n = 3 measurements, with error bars indicating the standard deviations of these values. The exponential regression for drone orbiting outdoors: time = 11.7 e−3.0m (R² = 0.9930). Data points shown are n = 1. Demonstration of the drone orbiting outdoors at height = 10 m, radius = 5 m, speed = 1.5 m/s around a fixed pole was performed at The Griffin Sportsground, flight site, London, SE21 7AL, United Kingdom.
A commercially available drone (DJI Mavic Air, 430 g, 168 × 184 × 64 mm) controlled using a remote controller connected to a smartphone application (DJI Go 4, iOS iPhone 7) was used for the tests. Modelling the critical process parameters of drone flight, the effects of temperature and vibration on insulin were investigated using the pharmacopoeia methods. The testing on human insulin has proven that insulin quality was maintained after exposure to environments that mimic a 30-minute drone delivery (temperatures −20 to +40 ◦C, vibration frequencies 0–40 Hz). Dynamic light scattering identified the active tetrameric and hexameric forms of insulin post testing. Vibration frequencies during drone flight were between 0.1 and 3.4 Hz.
Following drone transportation, there was no adverse impact on Actrapid apart from slight bubble formation post flight. The tests give supporting evidence that drone transportation of insulin is feasible from the point of view of pharmaceutical stability and maintenance of medicine quality. Hence, this research serves as a model for future studies investigating other types of medicines, environmental conditions or different drones.
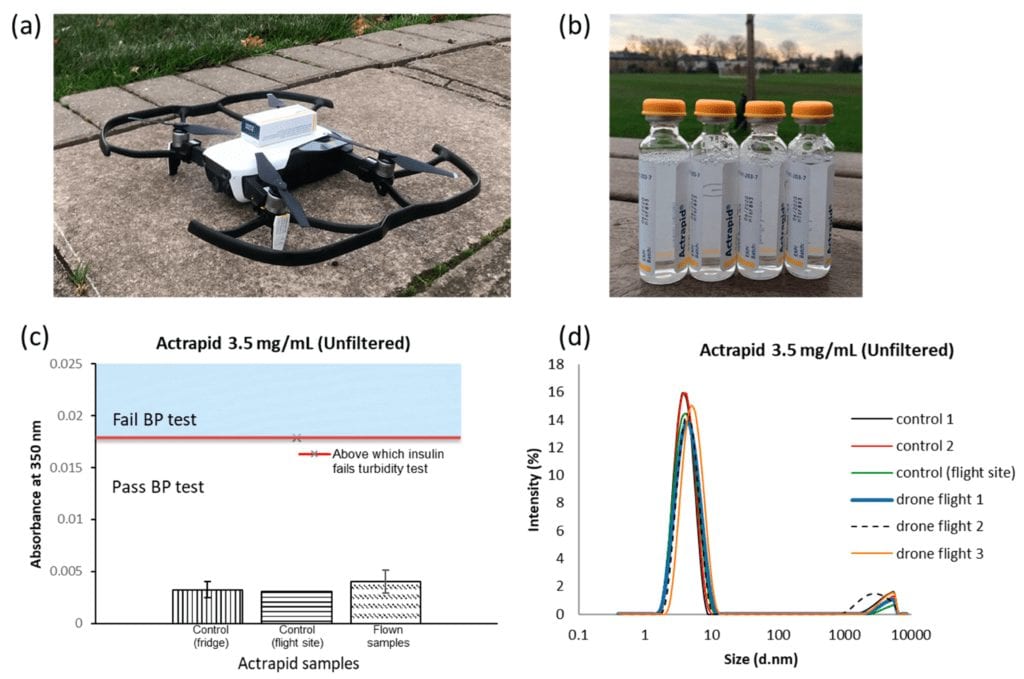
(a) Actrapid in its original packaging was securely attached onto the Mavic Air. (b) Visual appearances of Actrapid after flight (9 ± 2 minutes, n = 3 control on the left). (c) Mean absorbance at 350 nm plotted against the flown and not flown Actrapid. Data points shown are the mean of n = 2 measurements for control (fridge), n = 1 measurement for control (flight site) and n = 3 measurements for flown samples, with error bars indicating the standard deviations on these values. (d) Intensity (%) plotted against particle diameter, given in nm, (d.nm) for three flown Actrapid samples, one control (brought to flight site) and two controls (from fridge).
The researchers recommend five tests when considering the drone delivery of medicines. These tests must determine the safe flight time and range, the quality of the medicine post flight, the onboard conditions experienced by the medicine, the security of the drone supply chain and the effect of drone failure on both the medicine and the environment.
- Considering the gross weight of the medicines to be transported, the safe flight time and safe range need to be determined. Testing the edge-of-failure by taking into account the likely variance of environmental conditions is required for the selection of the most appropriate drone.
- A quality test for the medicine is required post delivery to ensure that the pharmaceutical product has arrived in a stable form. Ideally this should be non-destructive, simple to perform and if possible, be based on pharmacopeia recommendations and QbD considerations such as critical material quality attributes.
- On board monitoring of the medicines environment during drone flight is required to record if or when conditions have deviated from the manufacturer’s recommendations. Critical process parameters such as temperature, pressure, vibration frequency and g-force should be monitored.
- Ensuring the security of the medicines within the drone supply chain is required, for example on board anti-tamper monitoring and recipient authentication is required.
- Understanding the effect of drone failure during flight, considering both the consequences on the medicine and the environment is required.
The work presented here has successfully addressed the first three of these tests, future work and collaboration with drone manufacturers will allow the development of the last two.
Citatation: An Evaluation of the Delivery of Medicines Using Drones, Michelle Sing Yee Hii, Patrick Courtney and Paul G. Royall, Drones 2019, 3(3), 52; https://doi.org/10.3390/drones3030052, https://www.mdpi.com/2504-446X/3/3/52/htm